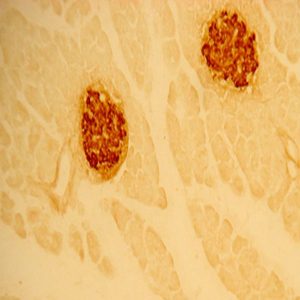
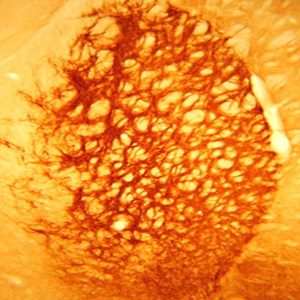
Description
The antibody has a proven strong biotin-streptavidin/HRP staining at a 1/2000 – 1/4000 dilution in rat brainstem, cerebellum and adrenal medulla.
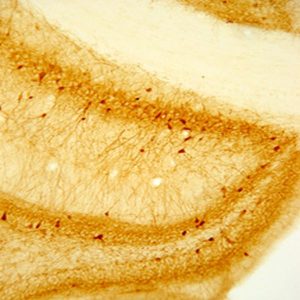
The above image is of DBH positive neurons in rat brainstem, vibratome sectioned, ABC / DAB detection.
Using Western blot of purified DBH the antiserum detects a triplet at approximately 72-74 kD. Optimal dilution will vary depending upon fixation, labeling technique and/or detection system; therefore, a dilution series is recommended.
Photo Description: IHC image of neurons staining for DBH in the rat brainstem. The tissue was fixed with 4% formaldehyde/0.05% glutaraldehyde in 0.1 M phosphate buffer, before being removed and prepared for vibratome sectioning. Floating sections were incubated at RT in 10% goat serum in PBS, before standard IHC procedure. Primary antibody was incubated at 1:4000 for 48 hours, goat anti-rabbit secondary was subsequently added for 1 hour after washing with PBS. Light microscopy staining was achieved with standard biotin-streptavidin/HRP procedure and DAB chromogen.
Host: Rabbit
Quantity / Volume: 100 µL
State: Lyophilized Whole Serum
Species Reactivity: Bird, Cat, Ferret, Finch, Guinea Pig, Hamster, Hatchetfish, Human, Ilyanassa Obsoleta (Sea Snail), Monkey, Mouse, Pig, Quail, Rat, Sparrow, Starling, Steer (Cattle), Turkey, Turtle, Zebra Finch
Availability: In Stock
Alternate Names: DBM; DOPO; Dopamine beta-monooxygenase, anti-DBH
RRID: AB_572229
Immunogen: Bovine DBH
Gene Symbol: DBH
Database Links:
Entrez Gene: 101082957 Cat
Entrez Gene: 101687995 Ferrett
Entrez Gene: 102038113 Finch
Entrez Gene: 100727873 Guuinea Pag
Entrez Gene: 100761411 Hamster
Entrez Gene: 1621 Human
Entrez Gene: 574105 Monkey
Entrez Gene: 13166 Mouse
Entrez Gene: 733609 Pig
Entrez Gene: 107321868 Quail
Entrez Gene: 25699 Rat
Entrez Gene: 102067155 Sparrow
Entrez Gene: 106851290 Starling
Entrez Gene: 280758 Steer (Cattle)
Entrez Gene: 100544779 Turkey
Entrez Gene: 102455557 Turtle
Entrez Gene: 100224356 Zebra Finch (Bird)
Technical Sheets
This product contains the preservative sodium azide. The concentration percent of the sodium azide is ≤ .09%. Although this hazardous substance is a concentration below that required for the preparation of a Material Safety Data Sheet, we created a standard MSDS for your records.
Download Data SheetDownload MSDS
Reviews
Want to leave a review? Please click here to send us your review.
KO validated in mouse brain
Antibody worked very well at 1:2000 for fluorescent IHC (free-floating).
40 um mouse brain sections (perfused, PFA fixed) were incubated overnight in 5% NGS/ 0.1% Triton-X PBS blocking serum. Brain sections at the level of the locus coeruleus (LC) from control and DBH KO mice were compared. Primary antibody binding was detected with goat anti-rabbit AlexaFLuor 488, and produced no signal in the LC of DBH KO animals but brightly labeled LC cell bodies in controls.
Submitted by Daniel Lustber, Emory University on 08/20/2021
DBH Antibody Worked Beautifully
Mouse hearts were perfused with PBS (pH 7.4) at RT, followed by cold fixative (4% paraformaldehyde + 0.2% picric acid in PBS). Tissues were fixed again for 2h at 4°C in cold 20% sucrose/PBS, and sectioned in a microtome. Slides was wrapped in aluminum foil and stored at −20°C.
Slide-mounted heart tissue sections were washed 3 x 5 min in 0.1M PBS, permeabilized with 0.4% Triton X-100 plus 0.5% BSA for 20 min. Tissues were then incubated overnight with DBH antibody (1:1000 dilution), washed four times with 0.1M PBS (10 min each), permeabilized with 0.4% Triton X-100 + 0.5% BSA, and incubated for 1.5h with a secondary antibody. After washing, labeled tissue sections were viewed under a confocal microscope system.
Labelled tissue sections showed clear labeling while the negative control sections (with our DBH antibody) showed only background fluorescence. This DBH antibody worked beautifully in our experiments!
Jason Zhang, Project Scientist
University of California- San Diego
Very nice antibody
We used 22806 in rat tissue.Brains were fixed using 4% paraformaldehyde, saturated in 30% sucrose and stored at -80. Brains were sectioned at 40 um using a freezing/sliding microtome and sections were stored in cryoprotectant at -20.
The antibody clearly labeled fiber innervation in the cortical areas and innervation was easily measured via stereology. Brains were incubated overnight at RT at 1:500 in TBST +1% Goat Serum after blocking in 10% Serum in TBST.
Vector biotinylated anti-rabbit was then used at a 1:500 dilution in TBST. After 1 hr ABC incubation, antibody was detected with DAB Nickel with a 10 minute development.Submitted by:
Sarah Kelly
PhD Candidate
Department of Translational Science and Molecular Medicine
Michigan State University
Very happy with the results
We used rabbit anti-bovine DBH (#22806) in an ICC Zenk protocol with sectioned zebra finch brain tissue and were very happy with the results. The antibody provided clear staining with minimal background. The antibody clearly labeled neuron bodies as well as tracks.
Our protocol followed Sockman & Salvante 2007. Zebra finch brains were fixed using 4% paraformaldehyde, saturated in 30% sucrose and stored at -80. Brains were sectioned at 40 um using a cryostat and sections were stored in cryoprotectant at -20.
Primary
1. 3×5 min wash in PBS
2. 15 min incubation in 0.1% sodium borohydride
3. 3×5 min wash in PBS
4. 30 min incubation in 0.5% H2O2 in PBS
5. 3×5 min was in 0.3% PBST
6. 1 hr incubation in 20% normal goat serum
7. 15 min incubation in Aviden solution
8. 3×5 min wash in 0.3% PBST
9. 15 min incubation in Biotin
10. 3×5 min wash in 0.3% PBST
11. 48 hr incubation at 4˚C at 1:16,000 in PBSTN
Secondary
1. 1 hour incubation in secondary antibody at RT
2. Visualize using ABC kit and DAB
3. Dehydrate and coverslip
Submitted by:
Kendra Sewall, PhD
Assistant Professor
Biological Sciences, Virginia Tech
Antibody Worked Very Well
We tried the rabbit anti-dopamine-beta-hydroxylase antibody in ferret tissues, more specifically on brainstem tissues. Ferrets were perfused with saline followed by 4% paraformaldehyde and postfixed for 12 hours.
We used 1:500 dilution of DBH antibody and stained with Alexa 594 as a secondary antibody. The citation of the antibody has been published in the following article:
Allergic lung inflammation affects central noradrenergic control of cholinergic outflow to the airways in ferrets. Wilson CG, Akhter S, Mayer CA, Kc P, Balan KV, Ernsberger P, Haxhiu MA. J Appl Physiol. 2007 Dec;103(6):2095-104. Epub 2007 Sep 13. PMID:17872402[PubMed]
Submitted by:
Prabha Kc, Ph.D.
Assistant Professor
Case Western Reserve University
Department of Pediatrics
Cleveland, OH
Detected small fibers with very little background
Animals were briefly anesthetized with pentobarbital (200mg/kg) followed by transcardial perfusion with 200 ml heparinized saline and 500 ml Zamboni fixative. Brains were then removed and stored in 30% sucrose until further processing.
IHC
Brains were frozen and cut at 50 µm thick sections using a cryostat.
Free-floating sections were first washed with PBS and then pretreated with a solution of 50% methanol, 0.3% hydrogen peroxide in PBS for 1 hour.
Blocking Solution: 1% triton-X 100, 2% fetal bovine serum in .1 M PBS for at least 1 hour at room temperature.
Dilutent for primary and secondary antibodies: 0.5% triton-X 100, 2% fetal bovine serum in .1 M PBS at
Primary dilution 1:5000 overnight at 4°C
Secondary Alexa 488 (Life Technologies) 1:500 in dilutent for 1 hour
Sections were washed thoroughly in .1 M PBS following detection, coverslipped and mounted with Vectashield mounting medium (Vector Labs)
This antibody detected small fibers within the cortex, central amygdala and various nuclei of the thalamus and hypothalamus with very little background.
Note: The time which animals spend under anesthesia will affect the level of expression in the thalamus/hypothalamus regions.
Review submitted by:
Vanessa M. Kainz
Research Associate
Department of Anesthesia and Critical Care Beth Israel Deaconess Medical Center
High quality antibody
I used the DBH antibody and the results showed very nice staining and no background.
Zebra finch brain tissue
Flash frozen and stored at -80 degrees
Sectioned at 20 um on cryostat and placed on superfrost plus slides.
Slides stored at -80 degrees until processing.
4% paraformaldehyde -15 min @ RT (room temperature)
3 X 5 min washes in 1X PBS @ RT
0.9% H2O2 in methanol-15 min @RT
3 X 5 min washes in 1XPBS @ RT
10% normal goat serum – 1 hour @ RT
Primary antibody incubation: overnight @ 4 degrees
1 ul/ml antibody
10% normal goat serum
1XPBS-TritonX
Secondary antibody – 90 min @ RT
Visualize using ABC kit (Vector) and DAB
Dehydrate and coverslip.
Review submitted by:
Juli S. Wade, PhD
Yu-Ping Tang, PhD
Neuroscience Program
Michigan State University