Description
The ImmunoStar neuronal nitric oxide synthase C-terminal antiserum was quality control tested using standard immunohistochemical methods. The antiserum demonstrates strongly positive labeling of rat hypothalamus, striatum, cortex and spinal cord using indirect immunofluorescent and biotin/avidin-HRP techniques. Recommended primary dilution is 1/8,000 – 1/12,000 in PBS/0.3% Triton X-100 – Bn/Av-HRP Technique.
By Western blot analysis of brain homogenates the antibody specifically labels a band of approximately 155 kD. Immuno-labeling is completely abolished by pre-adsorption with synthetic human nNOS (1419-1433) at 5 µg per mL of diluted antibody. No cross reactivity with other forms of NOS were observed.
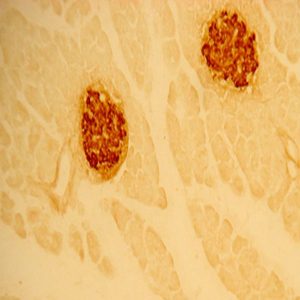
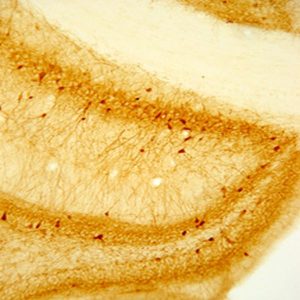
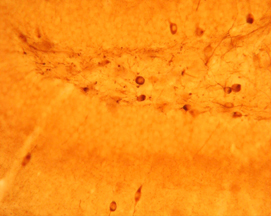
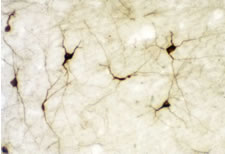
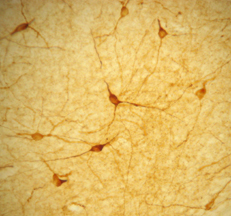
Photo Description: IHC image of a single neuron staining for the C-terminal of nNOS in the rat cortex (above), along with low magnification of cortex (below left), neurons in the rat dentate gyrus (below middle) and in the striatum (below right). The tissue was fixed with 4% formaldehyde in phosphate buffer, before being removed and prepared for vibratome sectioning. Floating sections were incubated at RT in 10% goat serum in PBS, before standard IHC procedure. Primary antibody was incubated at 1:10000 for 48 hours, goat anti-rabbit secondary was subsequently added for 1 hour after washing with PBS. Light microscopy staining was achieved with standard biotin-streptavidin/HRP procedure and DAB chromogen, below middle image with nickel stain.
Host: Rabbit
Quantity / Volume: 100 µL
State: Lyophilized Whole Serum
Reacts With: Cat, Chick, Ferret, Guinea Pig, Hamster, Human, Monkey, Mouse, Opossum, Rat, Shrew, Squirrel Monkey, Vole, Zebrafish
Availability: In Stock
Alternate Names: Constitutive NOS; NC-NOS; NOS type I; Neuronal NOS; Nitric Oxide Synthase; bNOS; Nitric oxide synthase-brain; N-NOS; Peptidyl-cysteine S-nitrosylase; IHPS1; NC-NOS; nitric oxide synthase 1 (neuronal), anti-nNos:C-Terminal
Gene Symbol: NOS1
RRID: AB_572256
Database Links:
Entrez Gene: 101100342 Cat
Entrez Gene: 427721 Chick
Entrez Gene: 101692439 Ferret
Entrez Gene: 100729682 Guinea Pig
Entrez Gene: 100758127 Hamster
Entrez Gene: 4842 Human
Entrez Gene: 693781 Monkey
Entrez Gene: 18125 Mouse
Entrez Gene: 100013712 Opossum
Entrez Gene: 24598 Rat
Entrez Gene: 102850179 Shrew
Entrez Gene: 101034544 Squirrel Monkey
Entrez Gene: 101984335 Vole
Entrez Gene: 60658 Zebrafish
Technical Sheets
This product contains the preservative sodium azide. The concentration percent of the sodium azide is ≤ .09%. Although this hazardous substance is a concentration below that required for the preparation of a Material Safety Data Sheet, we created a standard MSDS for your records.
Download Data SheetDownload MSDS
Reviews
Want to leave a review? Please click here to send us your review.
Great results with your antibody!
Our lab has used this to co-label NOS on mouse
neuroendocrine cells and cortex . We see neuronal staining at concentrations of
1:1000 (see green fluorescence in image below).
Below is our protocol:
Tissue prep: Perfuse transcardially with 15 mL 0.01 M phosphate-buffered saline (PBS)
followed by 20 mL of 4% paraformaldehyde (PFA) in 0.1 M phosphate buffer.
After 24 h post-fix in 4% PFA, cryoprotect in successive incubations of 15% and 30%
sucrose in PBS.
Immunofluorescent Labeling for cryosections: ImmnoStar NOS (Cat no24287) and
vasopressin antibody
Wash Tissue Sections
● Put hydrophobic pens around the tissue to keep the water from leaking off the
tissue.
● WASH PFA off sections with PBS X 3
Permeabilization and Block
● Diluent
o 5% normal donkey serum/normal donkey serum
o 5% BSA
o 0.3% Triton X-100
o Leave this diluent on for a total of 60 MINUTES.
● WASH with PBS X 3
Primary Antibody
● Make diluent
o 5% normal donkey serum/ normal donkey serum
o 5% BSA
o 0.03% Triton X-100
● Add antibodies at dilution below:
nNOS (1:1000 dilution; Immunostar; cat # 24287)
Vasopressin antibody
*Leave the slides in the cold room for 48-72 hours with parafilm coverslips in HUMID
chamber
2° Antibody
Vasopressin and nNOS colabel immunofluorescent labeling
● WASH with PBS X 3
● Diluent
o 1% BSA
o 2% NGS/NDS
o PBS
Adding the 2° antibody:
Incubated in Goat anti-rabbit Alexa Fluor 488, 1:1000, Life Technologies, USA and
donkey anti-mouse Alexa Fluor 594, 1:1000, Life Technologies, USA at room
temperature for 1.5 hours.
● WASH with PBS X 3
● Coverslip with Vectashield Hardset antifade mounting media with DPAI
(H-1400-10 Vector Laboratories, Burlingame, CA).

A go to antibody for PGC spinal motor neurons
My lab has used this antibody for many years to distinguish preganglionic motor neurons in the embryonic mouse spinal cord. It is very clean and concentrated. We generally use at a 1:10,000 dilution. An example can be found in one of our papers doi: 10.1016/j.neuron.2008.06.025 (see Fig 1U).
We process our spinal cord tissues by immersion in cold 4% PFA/PBS for 1.5-2 hours depending on the size/age of the sample, wash 3X with cold PBS, and cryoprotect in 30% sucrose before cryosectioning.
Antibody staining is achieved by thawing slides, overlaying with blocking solution (PBS, 0.1% TX100 , 1% Horse (or Donkey) serum) for 15 minutes. Then add primary antibodies diluted in the blocking solution. Incubate overnight at 4 degrees. Next day wash 3X with PBST (PBS with 0.1% TX100), and add fluorescent secondary antibodies (we generally use those from Jackson Immunoresearch) for ~1-2 hours at room temp. Wash 3X with PBST and coverslip using Prolong Gold (Invitrogen).
Fantastic antibody!
We have used this antibody to stain for nNOS positive neurons (also stains some processes) in frozen tissue sections and whole mount preparations of mouse intestine. We get consistent staining at 1:1500 although the staining of the neurons is so strong this can probably be dropped, even for quantification purposes!
Tissue prep:
4% PFA/PBS for 1.5 hrs, cryoprotected in 30% sucrose until they sink, OCT embedded, and sectioned.
For whole mounts, we open up intestine and fix in 4% PFA/PBS for 1.5 hrs.
IF protocol:
Sections:
Rehydrate in PBS, block with HINGS, stain in primary @4C O/N.
Wmts:
Wash in PBST, stain in primary with HINGS and DMSO @4C 48 hrs.
Sections:
Wash with PBS, stain with Alexafluor secondaries (Invitrogen), wash with PBS, and DAPI mount.
Wmts:
Wash in PBST, stain in secondary with HINGS and DMSO @RT 48 hrs.
Optional: proceed with MeOH dehydration and optical clearing.
Meenakshi Rao, MD PhD
Assistant Professor of Pediatrics
Columbia University Medical Center
New York, NY 10032