Description
The antibody has a proven maximum biotin-streptavidin/HRP staining at a 1/5,000 – 1/10,000 dilution in rat striatum, cortex, and hippocampus.
The antiserum has been characterized as specific to calbindin D-28k; please see reference listed below. Optimal dilution will vary depending upon fixation, labeling technique and/or detection system; therefore, a dilution series is recommended.
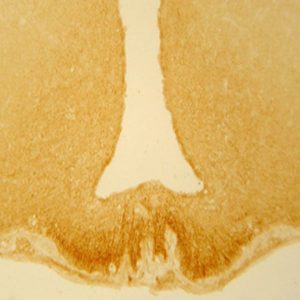
Photo Description: IHC image of neurons staining for the calbindin in the rat amygdala (above) and the rat cortex (below). The tissue was fixed with 4% formaldehyde in 0.1 M phosphate buffer, before being removed and prepared for vibratome sectioning. Floating sections were incubated at RT in 10% goat serum in PBS, before standard IHC procedure. Primary antibody was incubated at 1:10000 for 48 hours, goat anti-rabbit secondary was subsequently added for 1 hour after washing with PBS. Light microscopy staining was achieved with standard biotin-streptavidin/HRP procedure and DAB chromogen. The section was then mounted on slides with permount.

Host: Rabbit
Quantity / Volume: 100 µL
State: Lyophilized Whole Serum
Reacts With: Mouse, Rat
Availability: In Stock
Alternate Names: Calbindin; nCAB27; Calbindin D28; D-28K; Vitamin D-dependent calcium-binding protein; Vitamin D-dependent calcium-binding protein, avian-type; CALB1; calbindin 1, 28kDa, anti-Calbindin
RRID: AB_572222
Immunogen: Bovine Calbindin D-28K
Gene Symbol: CALB1
Database Links:
Entrez Gene: 12307 Mouse
Entrez Gene: 83839 Rat
NCBI Gene Aliases: MGC13895
Technical Sheets
This product contains the preservative sodium azide. The concentration percent of the sodium azide is ≤ .09%. Although this hazardous substance is a concentration below that required for the preparation of a Material Safety Data Sheet, we created a standard MSDS for your records.
Download Data SheetDownload MSDS
Reviews
Want to leave a review? Please click here to send us your review.
Core/Shell Differentiation
Our lab was able to use this for a beautiful differentiation of the Accumbens core and shell.
The protocol followed was essentially identical to the other review written for this product. However, we found that a dilution of 1:5000 was best for our purposes. It should be noted that we use a potassium ferrocyanide counterstain that seems to lighten Calbindin staining. Thus, my pilot studies would suggest that a dilution of 1:7000 would be sufficient for those that are not using such a counterstain.
The differences between 1:5000 and 1:7000 dilutions (as tested in increments of 500) were quite subtle, but did make some difference when trying to find the medial edge of the shell under the microscope. I would definitely recommend trying a range of dilutions for those attempting the same accumbens staining. Also, the DAB reaction was quite fast for the calbindin. We only needed 2-4 minutes before it seemed the reaction had reaches asymptote. Feel free to email me for a full protocol.
David Barker
http://rutgers.academia.edu/DavidBarker
Visualization of dorsolateral ventral pallidum
For our manuscript, Differential roles of ventral pallidum subregions during cocaine self-administration behaviors, in press at Journal of Comparative Neurology, we used the Immunostar rabbit anti-calbindin-d28k primary antibody to visualize the dorsolateral ventral pallidum subregion.
Rats were perfused with saline followed by 4% PF, stored in 30% sucrose, and coronally sectioned to 40 um. We followed the protocols of Zahm/Heimer and colleagues when they discovered the ventral pallidum subregions and their afferent/efferent projection patterns.
The protocol consisted of the following steps:
1. Wash in 0.1M phosphate buffer (PB; pH 7.4)
2. 15 min in 1% sodium borohydride
3. Wash
4. 1 hour blocking with PB containing 0.1% Triton X-100 and 3% normal goat serum
5. Primary antibody overnight at 4C – ImmunoStar rabbit anti-calbindin-d28k diluted 1 : 6000 in PB containing 0.1% Triton X-100 and 3% normal goal serum
6. Wash
7. 1 hour anti-rabbit biotinylated secondary (Vector) 1 : 200 in PB with 0.1% Triton X-100
8. Wash
9. 1 hour ABC (Vector) 1 : 200 in PB with 0.1% Triton X-100
10. Wash
11. Develop with 0.05% DAB for 6 min
Our study involved recording neurons within the ventral pallidum subregions during specific aspects of intravenous cocaine self-administration (approaching toward, responding on, or retreating away from a cocaine-reinforced operandum). In order to verify the placement of microwires within the VP subregions, prior to perfusion we passed current through each stainless steel microwire to leave an iron deposit at the uninsulated tip. After DAB (brown reaction) and mounting, we visualized the iron deposit by incubating in a 5% potassium ferrocyanide and 10% HCl solution (leaving a blue-green reaction).
For our purposes, this antibody strongly labeled fibers in dorsolateral VP with low background. Individual neurons and other fibers were clearly observed within distinct cortical layers that were not involved in our study.
David H. Root, Ph.D.
Rutgers University